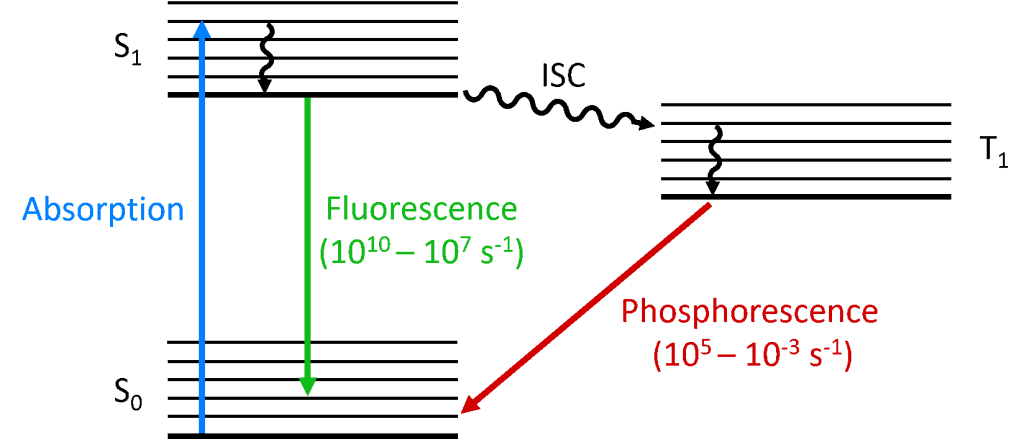
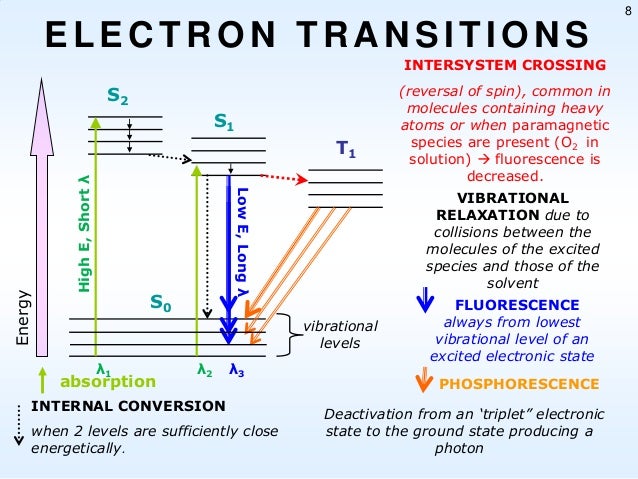
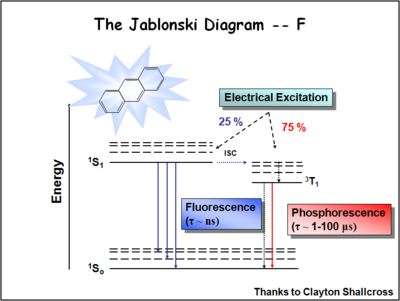
Fluorescence And Phosphorescence Diagram

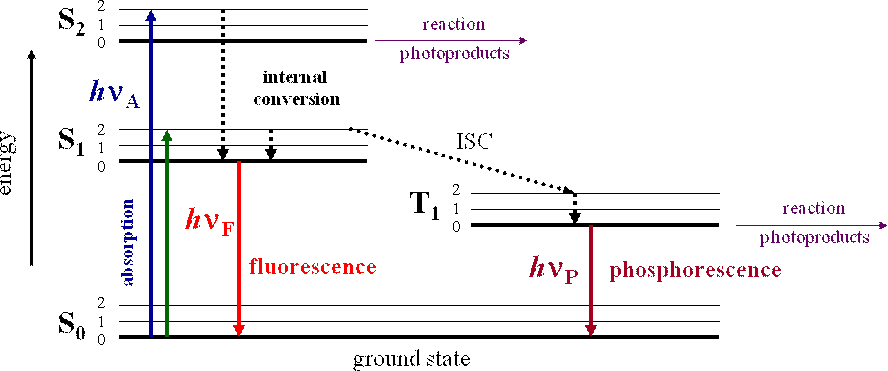
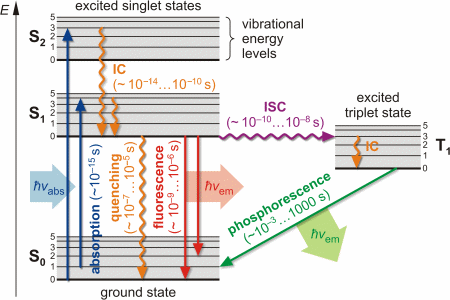
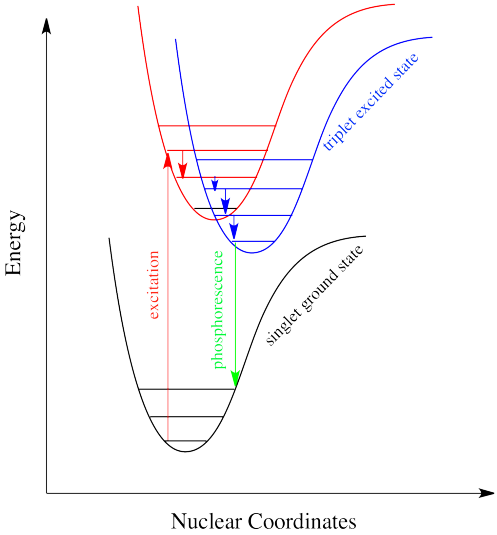
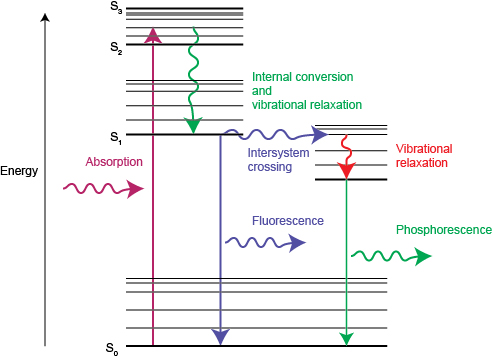
Interpretation of the energy diagram.
Fluorescence and phosphorescence diagram. Fluorescence de excitation can occur via a radiative decay ie. Introduction to fluorescence and the jablonski diagram. 7 differences between fluorescence and phosphorescence luminescence can be defined as the radiation emitted by a molecule or an atom on return to ground state from excited state after initial absorption of energy. A process in which a part of energy uv visible absorbed by a substance is released in the form of light as long as the stimulating radiation is continued.
Analytical instrumentation fluorescence phosphorescence measurement you may have come across terms like fluorescence and phosphorescence. By spontaneous emission of a photon. Fluorescence and phosphorescence. Unlike fluorescence a phosphorescent material does not immediately re emit the radiation it absorbs.
Than fluorescence or phosphorescence. Various parameters influence the emission of fluorescence. Possible de excitation pathways of excited molecules. Both fluorescence and phosphorescence are spontaneous emissions of electromagnetic radiation.
The radiative de excitation process can be described as a monomolecular process. It is a form of luminescence. The fluorescence emission took place from a singlet excited states average. The difference is that the glow of fluorescence stops right after the source of excitatory radiation is switched off whereas for phosphorescence an afterglow with durations of fractions of a second up to hours can occur 67.
Fluorescence and phosphorescence are two mechanisms that emit light or examples of photoluminescence. However the two terms dont mean the same thing and dont occur the same way. You may have a lot of questions in your head. In most cases the emitted light has a longer wavelength and therefore lower energy than the absorbed radiation.
Phosphorescence is a type of photoluminescence related to fluorescence.

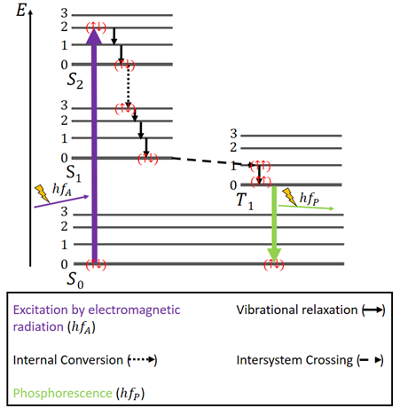

.png)
.png?revision=1&size=bestfit&width=580&height=392)