Fluorescence Microscopy Diagram

Fluorescence microscopy is a powerful tool for modern cell and molecular biologists and in particular neurobiologists.
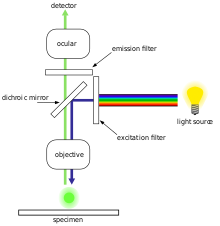
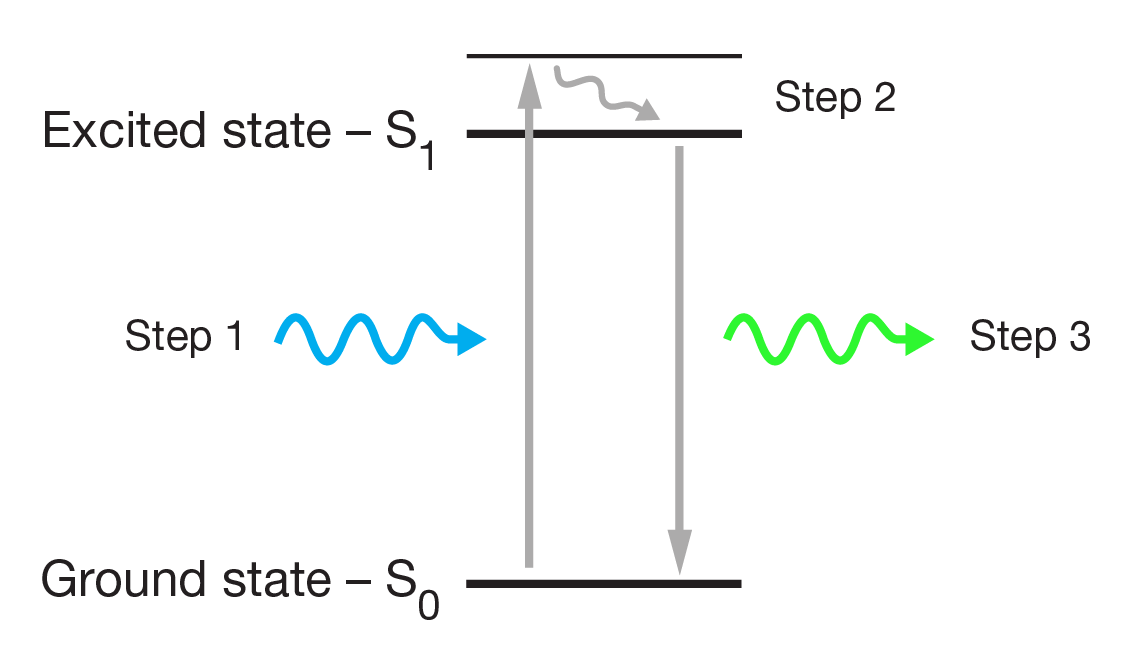
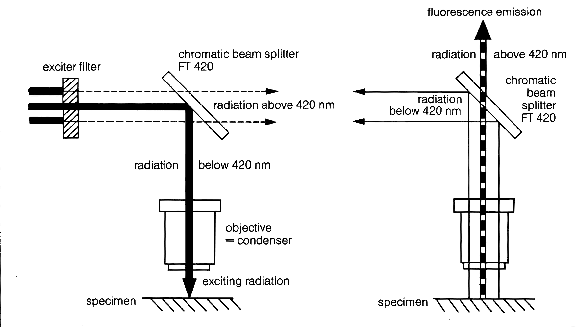
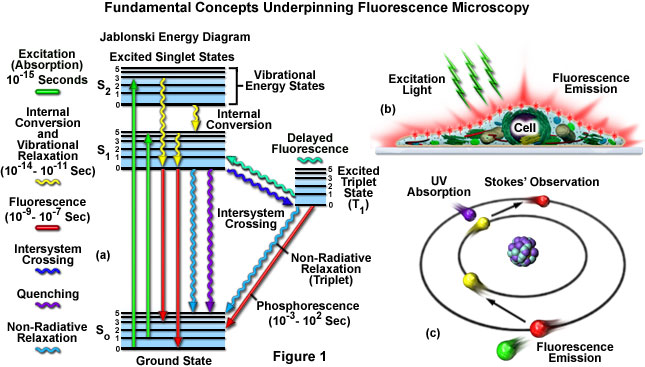
Fluorescence microscopy diagram. Fluorescence activity can be schematically illustrated with the classical jablonski diagram first proposed by professor alexander jablonski in 1935 to describe absorption and emission of light. A fluorescence microscope is an optical microscope that uses fluorescence and phosphorescence instead of or in addition to scattering reflection and attenuation or absorption to study the properties of organic or inorganic substances. Fluorescence is one of the most commonly used physical phenomena in biological and analytical microscopy mainly because of its high sensitivity and high specificity. It is a form of luminescence.
It provides a window into the physiology of living cells at sub cellular levels of resolution. In most cases the emitted light has a longer wavelength and therefore lower energy than the absorbed radiation. Find out how fluorescence microscopes support your research. The simplified diagram below shows absorption by molecules to produce either the first s1 or second s2.
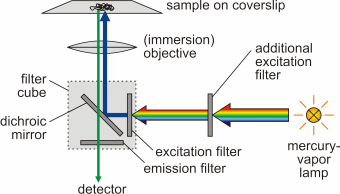
An introduction to fluorescence spectroscopy 7 fluorescence at room temperature most molecules occupy the lowest vibrational level of the ground electronic state and on absorption of light they are elevated to produce excited states. 1 cut away diagram of an upright microscope equipped both for transmitted light and epi fluorescence microscopy. The vertical the vertical illuminator in the center of the diagram has the light source at one end episcopic lamphouse and the filter cube at the other. Microscopes with an upright style frame are capable of producing fluorescence illumination either through episcopic or diascopic optical pathways although the latter is rarely used today.
Jablonski diagram for fluorescence an excited state electron rapidly on the order of 10 12 seconds loses its energy to vibration heat a process called internal conversion and falls to the lowest level of the first s 1 excited state.