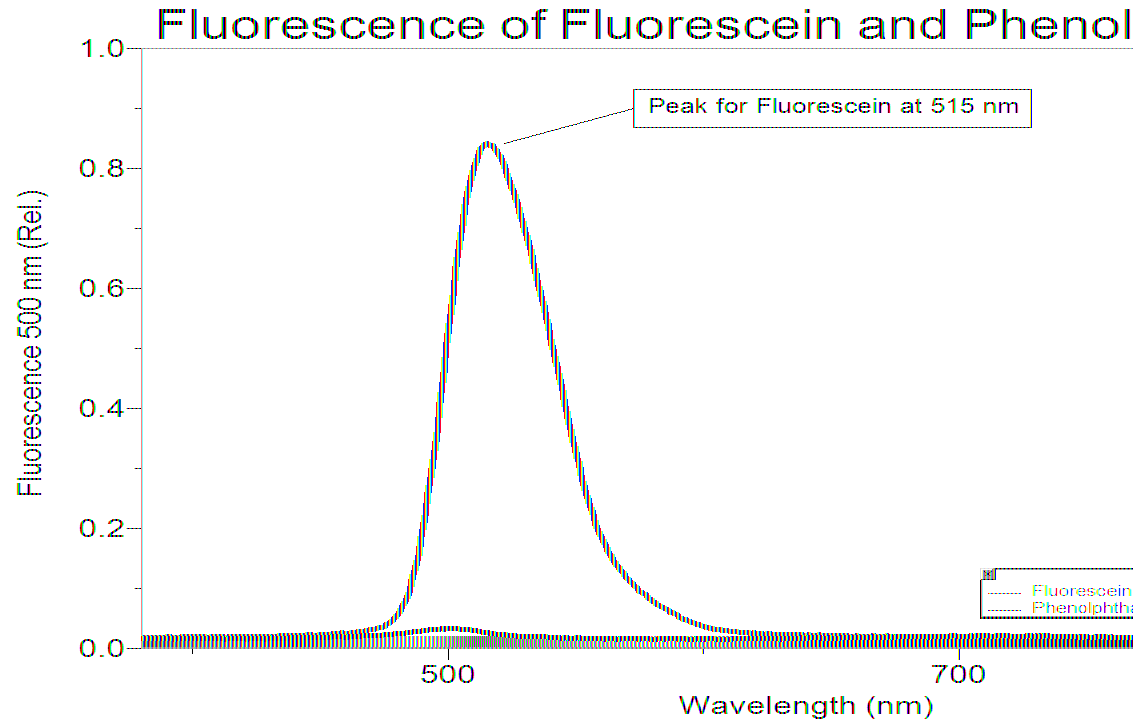
Fluorescence Spectroscopy Graph

Che425l fluorescence spectroscopy relative rate constants k x for the processes which deactivate the lowest excited singlet states namely fluorescence intersystem crossing external conversion.
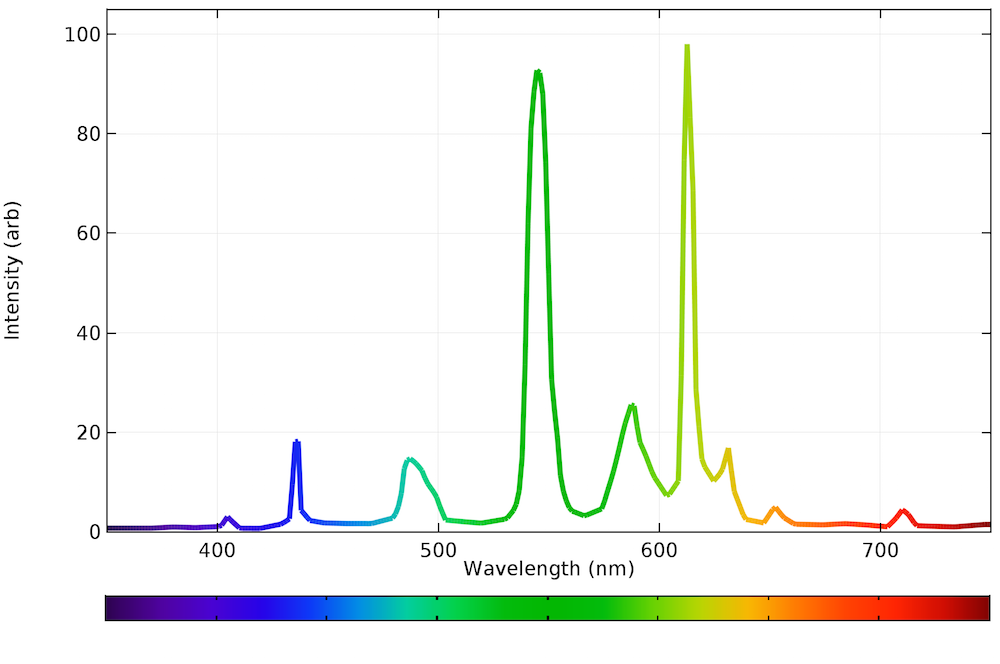
Fluorescence spectroscopy graph. Fluorescence spectroscopy fluorescence spectroscopy is an ideal method by which to study the interaction of the hp1b chromodomain with histone h321 the interaction is mediated via the methyl groups of lysine 9 of histone h3 which fit into a hydrophobic pocket on the surface of the chromodomain formed by the aromatic residues tyr 21 phe 45 and trp 42. Atomic fluorescence spectroscopy afs techniques are useful in other kinds of analysismeasurement of a compound present in air or water or other media such as cvafs which is used for heavy metals detection such as mercury. Fluorescence scanners including microarray readers resolve fluorescence as a function of spatial coordinates in two dimensions for macroscopic objects such as electrophoresis gels blots and chromatograms. This is a simulation of a photodiode array or rapid scanning spectrofluorometer.
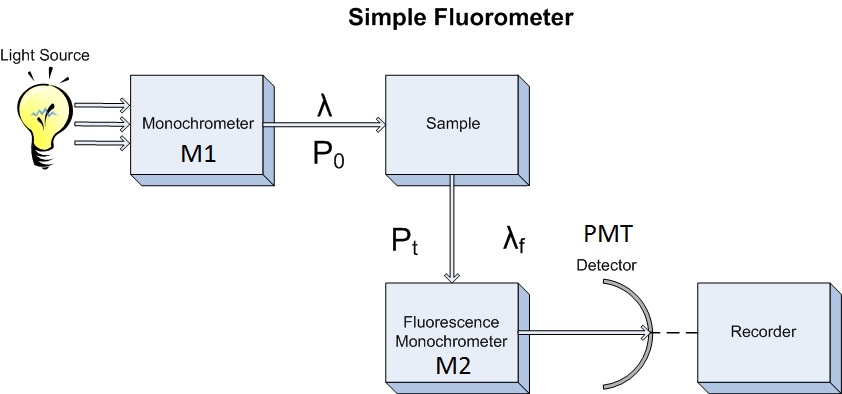
Fluorescence spectroscopy is a type of electromagnetic spectroscopy which analyzes the emission of light by a substance that has absorbed light or other electromagnetic radiation. Duetta can be used as a fluorometer as a uv vis nir spectrometer to measure absorbance or as an instrument that measures true molecular fingerprints which require the acquisition of fluorescence and absorbance correcting for ife in real time. In scanning fluorescence correlation spectroscopy sfcs the measurement volume is moved across the sample in a defined way. 1 basic principles of fluorescence spectroscopy 11 absorption and emission of light as uorophores play the central role in uorescence spectroscopy and imaging we.
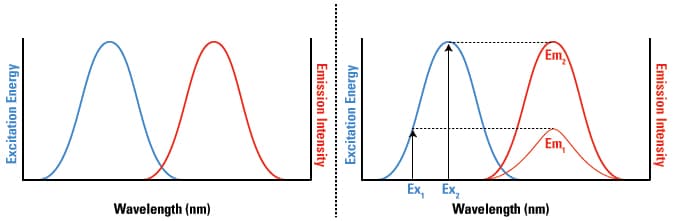
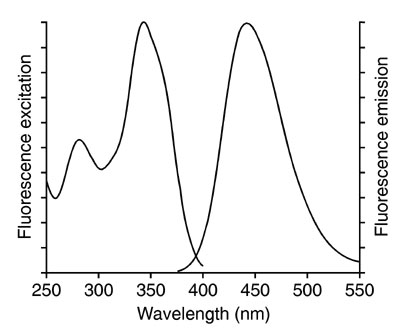
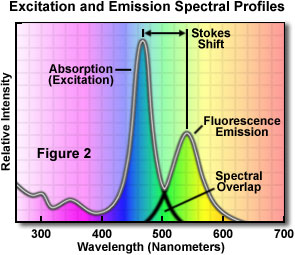
The simulation displays graphs of the excitation spectra emission spectra and synchronous spectra as well as a numerical value for the relative fluorescence emission intensity the black box labeled. An introduction to fluorescence spectroscopy 9 figure 2 idealised absorption and emission spectra in practice the 0 0 transitions in the absorption and emission spectra rarely coincide exactly the difference representing a small loss of energy by interaction of the absorbing molecule with surrounding solvent molecules.



.jpg)