Fluorescent Light Emission Spectrum
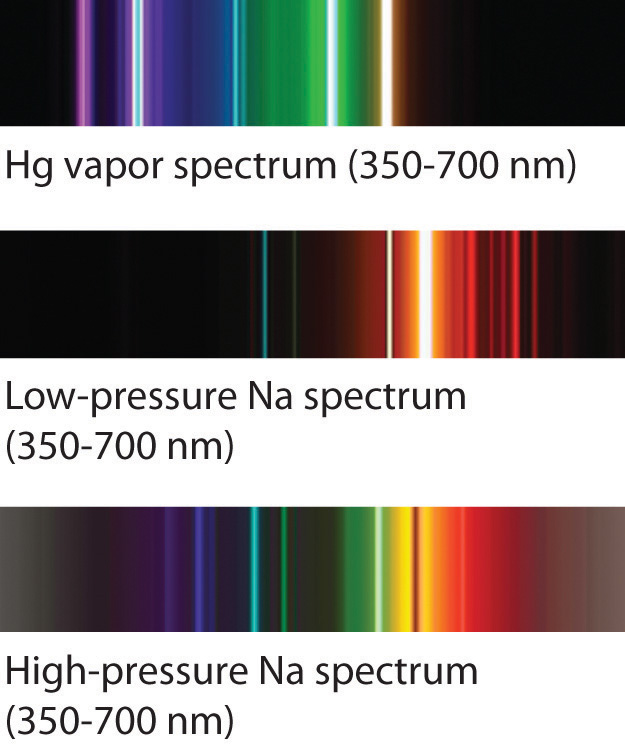
Fluorescence is the emission of light by a substance that has absorbed light or other electromagnetic radiation.
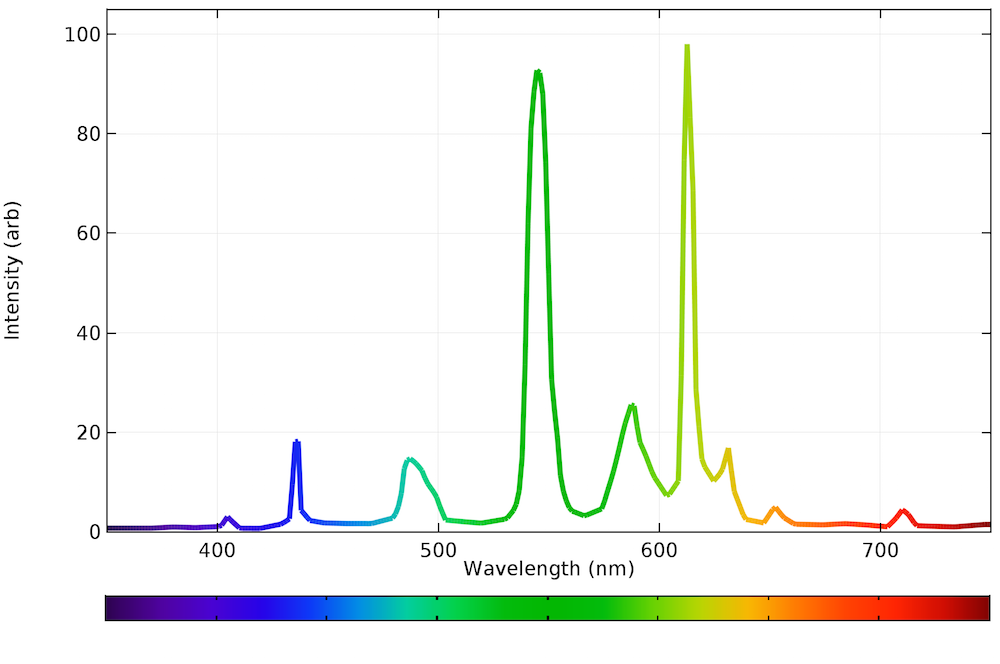
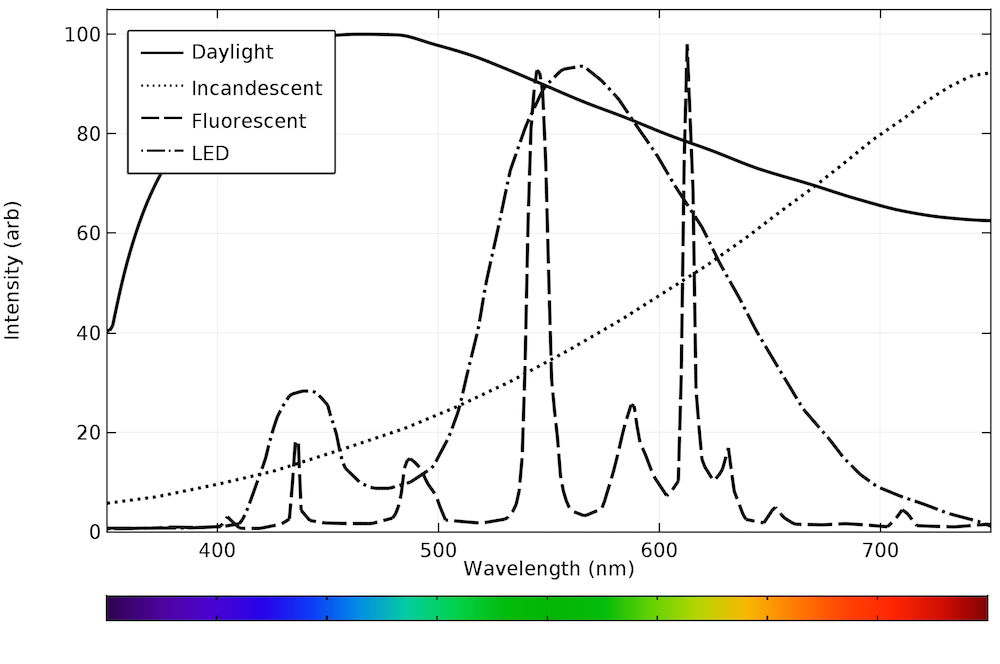
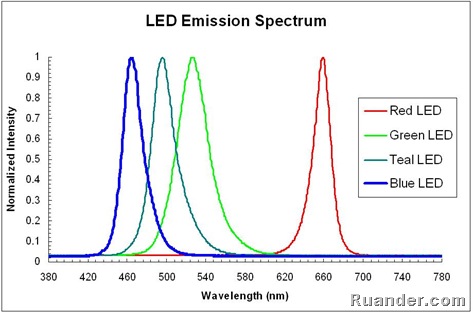
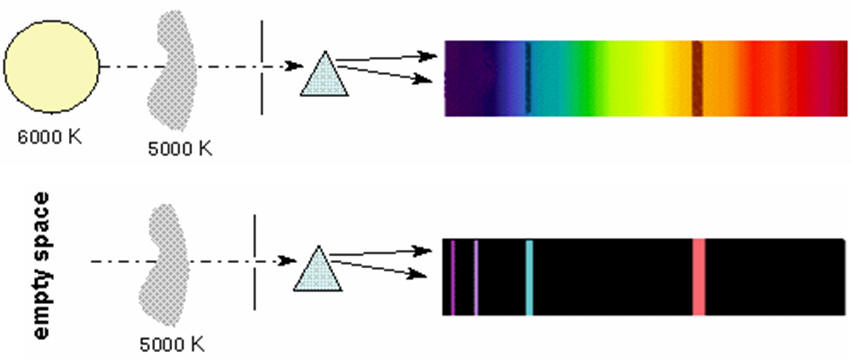
Fluorescent light emission spectrum. A fluorescent dye absorbs light over a range of wavelengths. How it works fluorescent light comes from two types of energy interactions. It is a form of luminescence. An excited fluorophore molecule emits lower energy light than the light it absorbs.
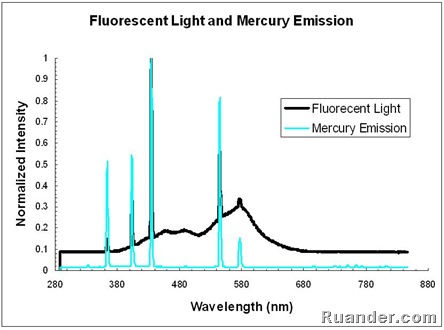
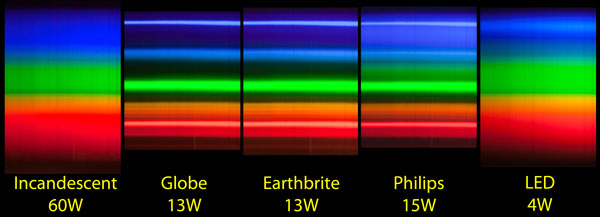
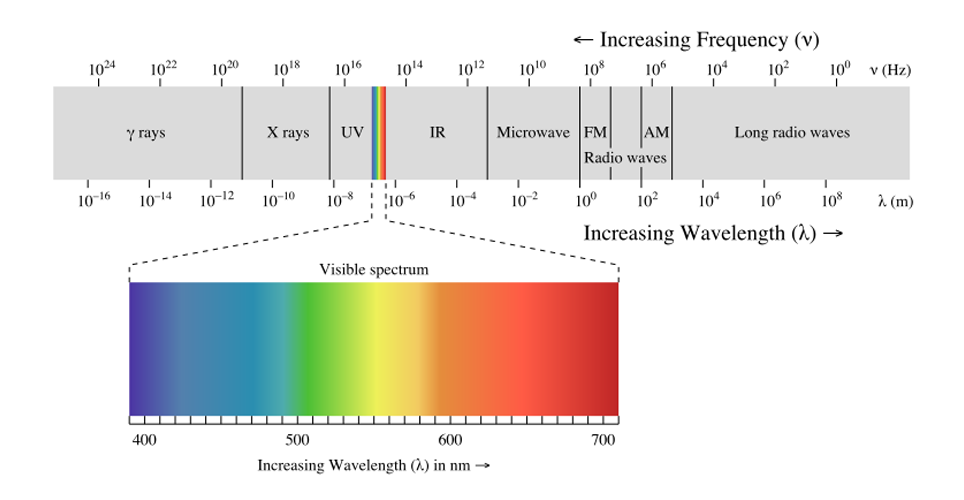
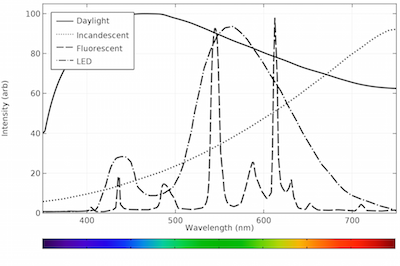
Effects change the spectrum and intensity of the emitted light and they must therefore be considered when analysing the emission spectrum of fluorescent light. The human eye can discern wavelengths between 400 and 700 nanometers which it interprets as a range of colors comprised of violet blue cyan green yellow orange and red. The emission spectrum for natural light generally follows the planck distribution in the visible part of the spectrum as we can see below. A look at how fluorescent lights are spectroscopically different from the old fashioned incandescent light bulb.
Fluorescence spectroscopy is primarily concerned with electronic and vibrational states. No color is dramatically favored over another although the intensity is highest in the light blue region around 460 nm. Therefore there is always a shift along the spectrum between the color of the light absorbed by the fluorophore during excitation and the color emitted. In most cases the emitted light has a longer wavelength and therefore lower energy than the absorbed radiation.
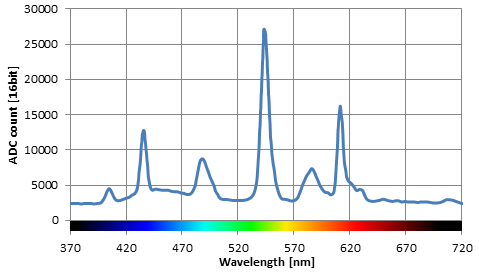
Spectrum of fluorescent light i bought a compact fluorescent light recently to replace an incandescent desk lamp. Popular taqman real time pcr assays antibodies oligos primers probes geneart gene synthesis cell culture plastics. The spectrum of visible light is a minuscule portion of the electromagnetic spectrum.